Two 52-year-old men sit across from me in the same week. Same chronological age. Same BMI. Similar exercise histories. One's biological age — measured by the GrimAge epigenetic clock — is 44. The other's is 61. One of them has the cellular machinery of a healthy 40-something. The other's DNA methylation patterns look like a man in his early 60s.
The difference matters enormously. GrimAge, the epigenetic clock trained specifically to predict mortality, is one of the strongest independent predictors of how long you'll live and how well you'll function as you age. And it's not captured by any of the biomarkers your primary care physician measures.
Chronological age tells you how many times you've orbited the sun. Biological age tells you how your cells are actually aging. They are often not the same number — and that gap is the most important health signal most people never receive.
What Is Biological Age, and How Is It Measured?
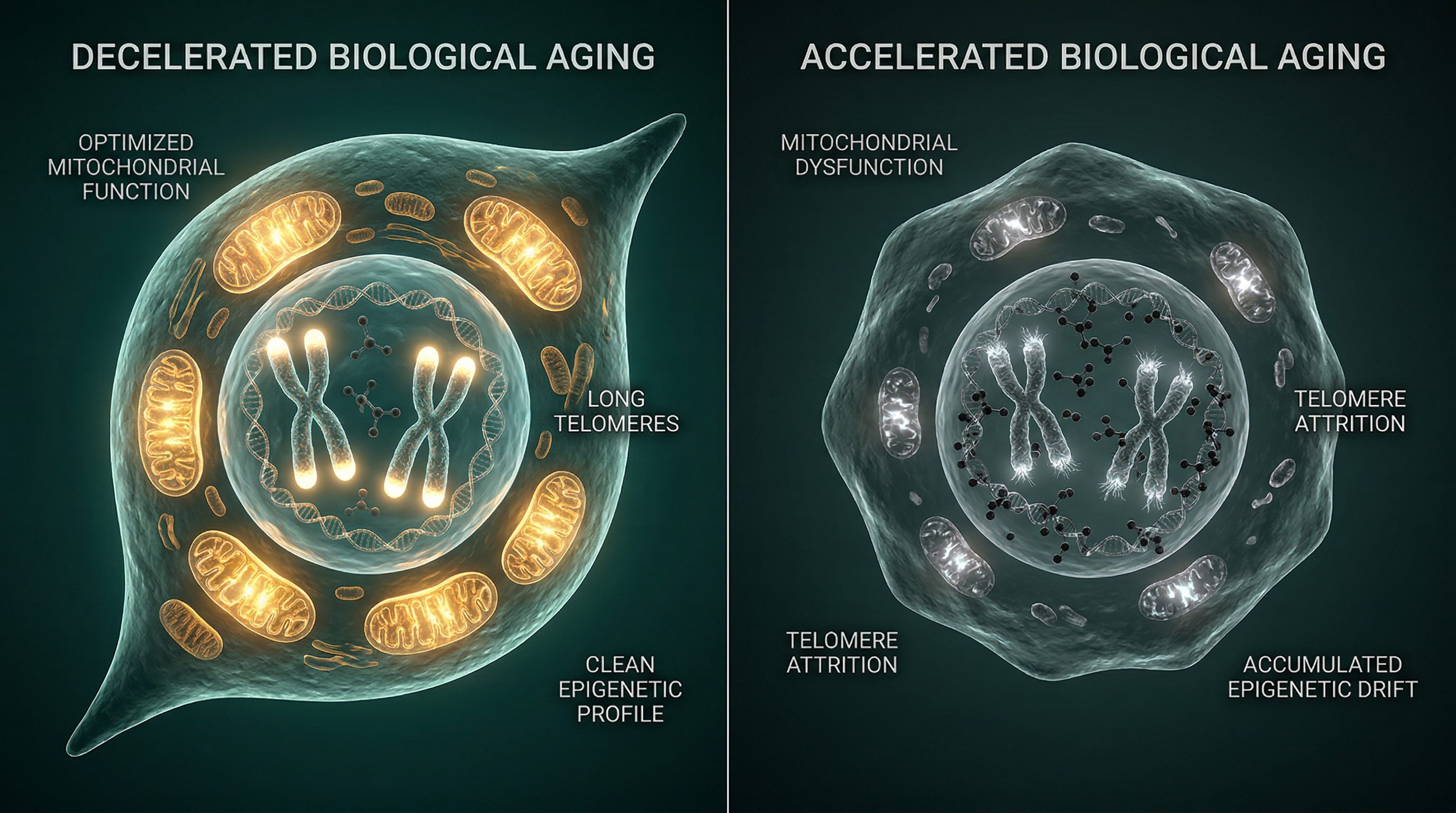
Biological age refers to the functional state of your cells and tissues — how well they're performing relative to what's typical for your chronological age. It's the accumulation of cellular damage, epigenetic drift, mitochondrial decline, and systemic inflammation playing out across decades of molecular history.
The most validated method for measuring it is the epigenetic clock — a mathematical model that reads DNA methylation patterns and predicts biological age. DNA methylation refers to chemical tags on your DNA that turn genes on and off without changing the underlying sequence. These tags change in highly predictable ways as we age, and deviations from expected patterns reflect either accelerated or decelerated biological aging.
Unlike your fasting glucose, A1c, or cholesterol panel, an epigenetic clock captures the cumulative biological effect of everything you've done and experienced — your diet, sleep quality, chronic stress, exercise history, smoking, environmental toxin exposures, and more — written into the chemical architecture of your DNA. It is, in many ways, the most honest biomarker in medicine.
Three Generations of Epigenetic Clocks
Not all epigenetic clocks are created equal. Understanding the generational differences is essential for anyone evaluating commercial testing options or interpreting clinical data — because the clock you measure matters as much as the measurement itself.
First-Generation Clocks: Horvath and Hannum
The first-generation clocks — developed by Steve Horvath and Gregory Hannum — were trained on chronological age. They correlate with actual age at levels exceeding 0.90, which is impressive. But correlating with chronological age is not the same as predicting health outcomes. Think of these clocks as a biological calendar: accurate at telling you where you are in time, but not particularly useful for forecasting where you're going. Horvath's clock alone is a weak predictor of mortality compared to second-generation alternatives.
Second-Generation Clocks: PhenoAge and GrimAge
The second generation shifted the training target from chronological age to health outcomes and mortality — and that shift changed everything. GrimAge, developed by Steve Horvath and Ake Lu, uses 1,030 CpG sites and integrates DNA methylation data with smoking history and inflammatory biomarkers to produce a mortality-calibrated biological age estimate.
GrimAge is sometimes called the "death clock" — not dramatically, but accurately. It is consistently the strongest methylation-based predictor of mortality across multiple large cohorts. A 2024 NHANES analysis of 2,105 adults followed for up to 20 years found that GrimAge epigenetic age acceleration was the only epigenetic clock that significantly predicted cardiovascular mortality — and among the strongest predictors of all-cause mortality. A 2025 Lancet Healthy Longevity study confirmed that GrimAge "consistently exhibits a positive association with frailty and higher baseline risk."
PhenoAge, the other prominent second-generation clock, was trained on a composite of clinical biomarkers. It is clinically useful but generally performs below GrimAge for mortality prediction in direct comparisons.
Third-Generation Clocks: DunedinPACE
DunedinPACE represents a conceptual leap. Rather than estimating a static biological age, it measures the pace of aging — how fast biological aging is progressing right now. A score of 1.0 means you are aging at exactly one year per calendar year. A score of 1.2 means you're aging 20% faster than the chronological clock. A score of 0.8 means you're aging 20% slower — compounding biological advantage over time the way a superior investment compounds returns.
DunedinPACE is derived from the Dunedin longitudinal cohort study in New Zealand, which followed participants across decades and identified the physiological changes that tracked with accelerated functional decline. A landmark 2026 Nature Aging study of 699 participants followed for up to 24 years found that longitudinal changes in epigenetic clocks predicted mortality independently of baseline epigenetic age — and that combined baseline-plus-rate-of-change models achieved a C-statistic of 0.808 for GrimAge v.2, one of the highest concordance values reported in this literature.
Why It Matters: The Clinical Evidence
GrimAge and Mortality Prediction
The evidence base for GrimAge as a mortality predictor has grown substantially in the past three years. The 2024 NHANES analysis — 2,105 adults, up to 20 years of follow-up — established GrimAge epigenetic age acceleration as the dominant methylation-based predictor of cardiovascular and all-cause mortality, outperforming first-generation clocks by a clinically meaningful margin. A January 2026 review in Frontiers in Molecular Biosciences synthesized findings across multiple cohorts and concluded that individuals with epigenetic age acceleration face higher risks of all-cause mortality, frailty, cognitive decline, and functional impairment — consistently, across populations, controlling for traditional risk factors.
What this means clinically: GrimAge is not a research curiosity. It is arguably the strongest single biomarker of mortality risk currently available to practicing physicians — and it is largely absent from standard care.
DunedinPACE and the Rate of Aging
DunedinPACE has been associated with cognitive dysfunction, incident dementia, and all-cause mortality. The 2026 Nature Aging study confirmed that the rate of change in epigenetic clocks over time predicts mortality independently of where you start — meaning a patient whose DunedinPACE is worsening year over year carries a different risk profile than one whose pace is stable or improving, even if their absolute biological ages are similar. A second 2026 Nature Aging study investigating tissue-specific DNA methylation clocks found that organ-level epigenetic signals may capture pathology relevant to specific disease processes — suggesting the field is moving toward precision organ-level aging assessment.
What Epigenetic Clocks Are Actually Measuring
Approximately 28% of the human genome shows age-related methylation changes — a remarkable fraction, reflecting the scale at which epigenetic architecture reorganizes over a lifetime. The methylation patterns captured by epigenetic clocks are not random noise. They reflect the accumulated biological history of the organism: chronic psychological stress, decades of smoking, diet quality, sleep architecture, exercise volume and intensity, environmental toxin burdens, and the cumulative inflammatory load carried across years.
This has two important implications. First, biological age is not an indictment of genetics — it is largely a record of lived experience at the cellular level. Second, and far more important clinically: epigenetic age is modifiable. It is not a fixed read-only value encoded at conception.
Interventions with documented effects on epigenetic clocks include caloric restriction (multiple human trials), resistance and aerobic training, pharmacological agents (metformin and rapamycin in research contexts), sleep optimization, and smoking cessation. The magnitude of effects varies — and the translation of methylation shifts to proportional mortality reduction is still being quantified — but the directionality is consistent: interventions that reduce biological burden of disease tend to move epigenetic clocks in a favorable direction.
"The epigenetic clock is a dynamic indicator, not a fixed read-only value. Your cellular age responds to how you live."
The Limitations Clinicians Should Know
I want to be direct about the limitations of current epigenetic testing. Acknowledging them is not a reason to avoid testing — it's a reason to interpret results with appropriate clinical sophistication.
1. The 450K array problem. Most commercial epigenetic age tests measure between 450,000 and 850,000 CpG sites using microarray technology. Whole-genome bisulfite sequencing can measure all approximately 28 million CpG sites in the human genome. The methylation patterns captured by commercial arrays are a well-chosen subset — but they are not the complete picture. Current commercial tests are not the final word on biological age; they are the best currently available approximation.
2. Tissue specificity. Blood-based epigenetic clocks measure methylation in circulating blood cells. Your blood cell methylation patterns may not fully reflect the aging state of your brain, heart, liver, or kidney — tissues that are inaccessible for routine sampling. Organ-specific clocks are in active development, and the 2026 Nature Aging tissue-specific clock data suggests this will be an important frontier. For now, blood-based clocks are the practical standard.
3. Intervention interpretability. We know that certain interventions shift epigenetic clock scores. We do not yet fully understand whether a 2-year improvement in GrimAge translates directly to a 2-year reduction in mortality risk. The relationship is likely positive and clinically meaningful, but the translation coefficient has not been precisely established. This is an active area of research, not a resolved question.
4. Clock selection matters. First-generation clocks — Horvath, Hannum — are significantly weaker mortality predictors than GrimAge or DunedinPACE. If you or your physician are evaluating commercial testing options, ask specifically which clocks are being measured. A panel that reports only Horvath clock results is providing incomplete and less clinically useful information than one that includes GrimAge and DunedinPACE.
The Gap in Standard Care
Conventional medicine has no biomarker for the rate of cellular aging. Annual physicals don't measure it. Standard blood work doesn't capture it. The closest proxies — telomere length, inflammatory markers like hsCRP and IL-6, metabolic markers like fasting insulin — are crude approximations compared to methylation-based clocks. They capture fragments of the picture. Epigenetic clocks integrate them.
The gap matters because biological age acceleration is both detectable and treatable years before clinical disease emerges. A 52-year-old whose GrimAge reads 61 has time to intervene — if he knows about it. He can address the driving factors: chronic sleep deprivation, insulin resistance, inflammatory load, stress physiology. Without testing, that signal is invisible until it becomes a cardiac event or cancer diagnosis. The epigenetic clock converts an invisible risk into a measurable, trackable, and modifiable target.
This is the fundamental promise of precision longevity medicine: not reacting to disease after it declares itself, but detecting the trajectory years in advance and redirecting it.
How We Use Epigenetic Testing at Pravida Health
Epigenetic age testing is available across our membership tiers and is integrated with our broader longevity assessment: metabolic biomarkers, DEXA body composition, VO2 max, and comprehensive bloodwork. We focus on second-generation (GrimAge) and third-generation (DunedinPACE) clocks for clinical utility — the first-generation clocks are available but provide limited additional actionable information beyond what the more predictive clocks deliver.
At baseline, we use the epigenetic panel to identify areas of epigenetic age acceleration and the biological systems most likely responsible. A patient with elevated GrimAge driven primarily by inflammatory pathway methylation signatures gets a different intervention conversation than one whose acceleration appears metabolic in origin. The specificity matters — it points us toward targeted intervention rather than generic longevity recommendations.
For example: a 50-year-old whose GrimAge is 4 years older than his chronological age and who also carries elevated hsCRP and IL-6 is a candidate for aggressive anti-inflammatory protocol, dietary restructuring, and sleep apnea evaluation. A 50-year-old whose GrimAge is 3 years younger than chronological age with well-controlled metabolic markers gets a different conversation — about maintaining trajectory and monitoring for drift.
We retest at 12-month intervals as the standard cadence, though patients in active intervention programs sometimes test at 6 months to capture rate-of-change data. As the 2026 Nature Aging data shows, the trajectory over time is as informative as the absolute score — and in some respects more so.
What You Can Do Today
1. Reduce modifiable epigenetic risk factors. Smoking, chronic sleep deprivation, insulin resistance, and chronic psychological stress are the strongest documented drivers of epigenetic age acceleration. These are not abstract risks — they are directly readable in methylation data. If you are carrying any of these, they are aging your cells faster than your chronological clock.
2. Optimize sleep architecture. Sleep is the single most potent epigenetic intervention most people are not taking seriously. Consistent 7–8 hours per night, a dark and cool sleep environment, and active management of sleep apnea if present. The research on sleep and epigenetic aging is not ambiguous — chronic sleep restriction is one of the most reproducible drivers of biological age acceleration.
3. Implement Zone 2 aerobic exercise. Zone 2 — sustained moderate-intensity cardio at roughly 60–70% of maximum heart rate — is one of the most consistently documented exercise modalities for reducing biological age acceleration. It improves mitochondrial function, reduces inflammatory signaling, and enhances metabolic flexibility in ways that directly address epigenetic aging mechanisms.
4. Manage chronic inflammation. hsCRP and IL-6 are partial mediators of epigenetic age acceleration — meaning they are not just markers, but likely drivers. Anti-inflammatory nutrition, omega-3 supplementation at therapeutic doses, management of occult chronic infections, and periodontal health are actionable levers most people haven't optimized.
5. Consider an epigenetic age test. Commercial options include TruMe, Elysium, and Tally Health. Before testing, understand which clocks each platform measures and what their limitations are. A test reporting only first-generation clock results is less clinically useful than one reporting GrimAge and DunedinPACE. If you are working with a physician, integrate the results into your broader biomarker picture rather than interpreting them in isolation.
Frequently Asked Questions
What is the difference between biological age and chronological age?
Chronological age is simply how many years you've been alive. Biological age reflects the functional state of your cells and tissues — how much molecular damage has accumulated and how well your systems are operating relative to what's typical for your age. The two often diverge, and biological age is a stronger predictor of health outcomes and mortality.
What is the GrimAge epigenetic clock?
GrimAge is a second-generation DNA methylation clock trained specifically to predict mortality. It integrates methylation at 1,030 CpG sites with clinical biomarkers including smoking history and inflammation markers. GrimAge epigenetic age acceleration is one of the strongest currently available predictors of all-cause and cardiovascular mortality.
What is DunedinPACE?
DunedinPACE is a third-generation epigenetic clock that measures the pace of biological aging rather than a static biological age. A score of 1.0 means you're aging at exactly one year per chronological year. Scores above 1.0 indicate accelerated aging; below 1.0 indicates slower-than-expected biological aging. It is derived from the Dunedin longitudinal cohort in New Zealand.
Can biological age be reversed?
Epigenetic clocks are dynamic — they respond to lifestyle changes, metabolic optimization, and pharmacological interventions. Research has documented biological age reductions following caloric restriction, exercise programs, sleep optimization, and certain medications. However, whether these methylation shifts translate proportionally to mortality reduction is still being established.
How often should I get an epigenetic age test?
Annual testing is the most practical interval for tracking trajectory — though 6-month testing is sometimes used when an active intervention program is underway. The change over time is as informative as the absolute score.
Understand Your Biological Aging Rate
Epigenetic age testing is available across our membership tiers at Pravida Health. We use GrimAge and DunedinPACE alongside comprehensive metabolic, genomic, and functional assessments to build a complete picture of your biological trajectory — and a protocol to address it.
Schedule a Consultation View Memberships