Your last cholesterol panel told you LDL was 118 mg/dL. Total cholesterol 192. HDL 54. Your doctor said: "Great numbers — keep doing what you're doing."
Here's what the standard lipid panel didn't measure: how many atherogenic particles are actually circulating in your blood, damaging your arterial walls. LDL cholesterol is the concentration of cholesterol carried in LDL particles — not the number of particles. And particle number is what kills you, not cholesterol concentration alone.
The two tests that fix this problem — ApoB and Lp(a) — are inexpensive, widely available, and in March 2026, formally endorsed for broader testing by a landmark joint guideline from the American College of Cardiology and the American Heart Association. They are still not ordered on standard lipid panels.
If you've never had your ApoB or Lp(a) measured, you have incomplete cardiovascular risk information. Full stop.
What Is ApoB, and Why Does It Matter More Than LDL-C?
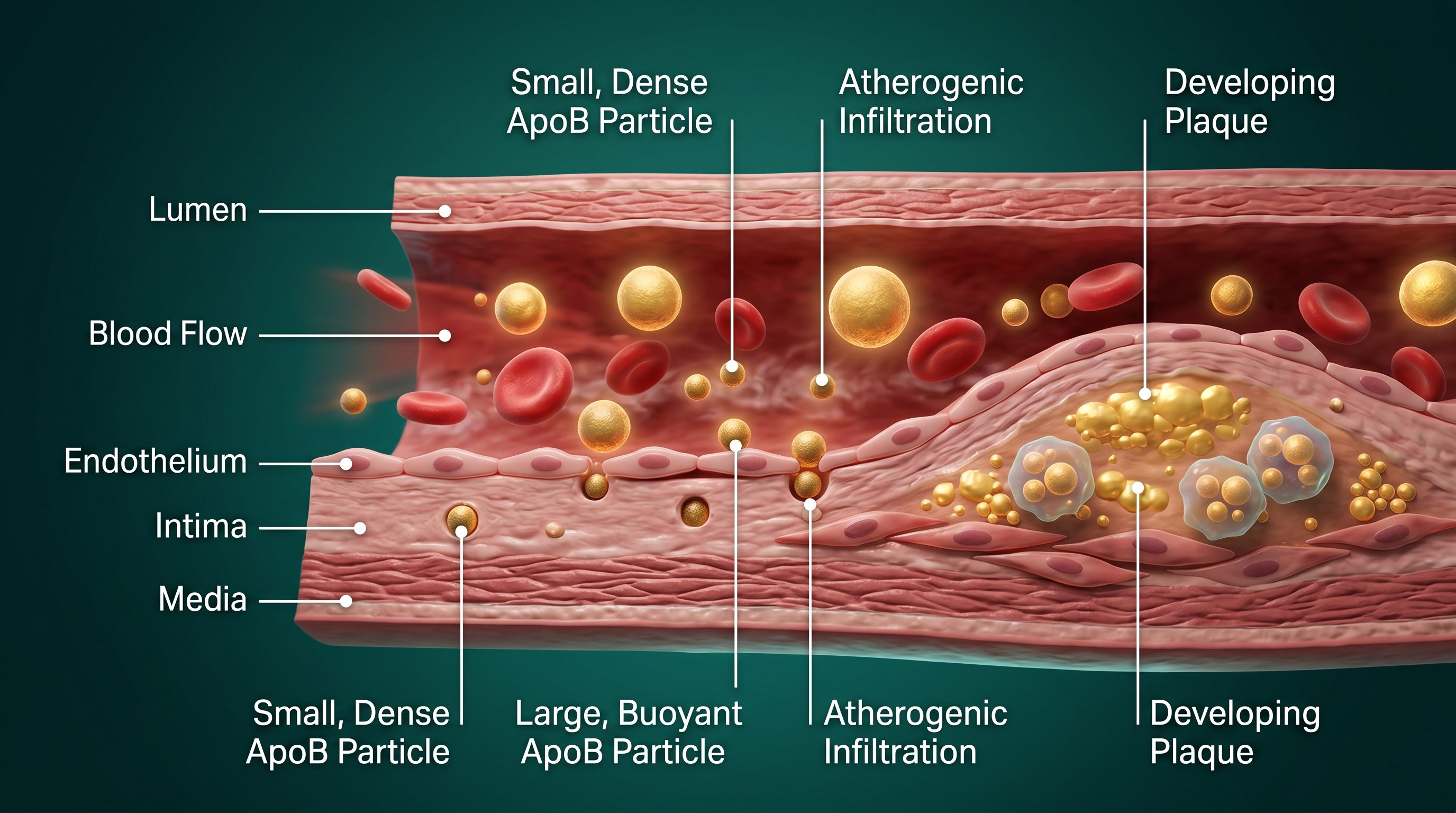
Apolipoprotein B (ApoB) is a protein on the surface of every atherogenic lipoprotein particle: LDL, VLDL, IDL, and Lp(a). One ApoB molecule per particle. This means ApoB is a direct count of the total number of atherogenic particles in your bloodstream — not an estimate, not a surrogate, but an actual particle count.
LDL cholesterol (LDL-C) measures the total amount of cholesterol carried inside LDL particles. The problem: particle size varies considerably from person to person. A patient with many small, dense LDL particles can carry a completely normal LDL-C while having an elevated ApoB — and correspondingly elevated cardiovascular risk that the standard panel will entirely miss. Conversely, a patient with few large, buoyant LDL particles may show a high LDL-C but carry a normal ApoB and lower actual particle burden.
This discordance — when LDL-C and ApoB tell different stories — is the clinically critical gap. It is not rare. It is common, particularly in patients with insulin resistance, elevated triglycerides, metabolic syndrome, and type 2 diabetes — the exact populations most likely to already carry elevated cardiovascular risk for other reasons.
The biology is straightforward: atherogenesis is driven by the number of apoB-containing particles that penetrate the arterial intima, not the cholesterol concentration those particles carry. More particles means more intimal penetration, more oxidized LDL, more macrophage activation, more foam cell formation, and ultimately more plaque. Counting particles — which is exactly what ApoB does — is more mechanistically direct than measuring cholesterol concentration.
The Evidence: ApoB Outperforms LDL-C
This is not a minority view in preventive cardiology. The evidence base has been building for two decades and was comprehensively synthesized in 2025.
A 2025 systematic review and discordance analysis published in the European Journal of Preventive Cardiology compiled 15 studies covering 593,354 participants across multiple populations and study designs. The headline finding: ApoB outperformed LDL-C in 9 of 9 studies where discordance was formally analyzed. In 7 of 9 studies where ApoB was compared to non-HDL-C — the next best proxy and the measure often recommended as an improvement over LDL-C alone — ApoB was significantly more accurate. The authors' conclusion was unambiguous: "neither LDL-C nor non-HDL-C are adequate clinical surrogates for apoB."
A separate 2025 UK Biobank study extended the finding further: ApoB outperformed even LDL particle number (LDL-P) for cardiovascular risk prediction when the two measures were discordant — with risk elevation detectable at discordance levels as small as 2%. This matters because LDL-P has sometimes been positioned as the superior alternative to LDL-C. The data suggest ApoB, which captures all atherogenic particles rather than just LDL particles specifically, is the more comprehensive risk measure.
These are not small studies with methodological problems. These are large, well-designed analyses. The consistent direction of the evidence is hard to argue with.
The 2026 ACC/AHA Guideline Change
In March 2026, a joint clinical guideline from the American College of Cardiology, American Heart Association, and nine other medical associations was published simultaneously in JACC and Circulation. The guideline addressed cardiovascular risk management across a broad set of patient populations and made several key changes that directly affect how we should be interpreting and ordering lipid panels.
The central change for ApoB: it may now be used to assess residual ASCVD risk and guide treatment in patients with cardiovascular-kidney-metabolic syndrome, type 2 diabetes, elevated triglycerides, or established cardiovascular disease who have already reached their LDL-C goals. The guideline explicitly states that "apoB may be a more accurate risk marker than LDL-C in these patient populations."
Read that carefully. The guideline is not saying ApoB is marginally interesting. It is saying ApoB may be a more accurate risk marker — in the specific populations where standard care would otherwise tell you the lipid panel is under control. These are exactly the patients who most need a more accurate measure.
The clinical implication is direct: if you have any metabolic complexity — insulin resistance, elevated triglycerides, diabetes, prediabetes, metabolic syndrome, or any established cardiovascular disease — ApoB is now guideline-supported as the more appropriate risk metric. Practicing medicine with only LDL-C in these patient populations is, as of 2026, no longer consistent with current guidelines.
The evidence-based ApoB targets by risk category:
General population: ApoB below 100 mg/dL
High cardiovascular risk (diabetes, metabolic syndrome, multiple risk factors): below 80 mg/dL
Very high risk / secondary prevention (established ASCVD, recent ACS): below 60–70 mg/dL
What Is Lp(a), and Why Is It Different From Everything Else?
Lipoprotein(a) — Lp(a) — is a distinct lipoprotein: an LDL-like particle bound to apolipoprotein(a), a large glycoprotein with structural homology to plasminogen. Unlike most cardiovascular risk factors, Lp(a) is not substantially modified by diet, exercise, statins, or most other interventions. It is approximately 80–90% genetically determined. Your Lp(a) level is essentially set by your genetics and does not change meaningfully across your lifetime in response to lifestyle choices.
This makes it simultaneously the most important and the most frustrating cardiovascular risk factor I encounter in clinical practice. Important because elevated Lp(a) represents significant, lasting cardiovascular risk that cannot be lifestyle-managed away. Frustrating because for most of the history of lipid medicine, there was nothing specific to do about it beyond aggressive management of every other controllable risk factor. That clinical reality is beginning to change — but more on that below.
The 2026 ACC/AHA guideline made universal Lp(a) testing formal: the guideline recommends testing at least once in adulthood for all adults. The reasoning is straightforward: approximately 20% of the global population — roughly 64 million Americans — have Lp(a) above 50 mg/dL (125 nmol/L), the threshold associated with approximately 1.4-fold increased long-term risk of myocardial infarction or stroke. Above 250 nmol/L, the risk approximately doubles. Most of these people have no idea their Lp(a) is elevated, because it is not measured on standard lipid panels. They may be receiving reassurance about their cardiovascular risk that is simply wrong.
The Mechanism
Lp(a) promotes atherosclerosis through two distinct, simultaneous pathways — which is part of why it is so clinically significant. First, it carries atherogenic cholesterol like other apoB-containing particles: it penetrates the arterial intima, oxidizes, and contributes to plaque formation through the same basic mechanism as LDL. Second, and more distinctively: apolipoprotein(a) is structurally homologous to plasminogen, the protein that dissolves blood clots. This means Lp(a) competes with plasminogen for binding sites, impairing normal fibrinolysis and promoting a pro-thrombotic state on top of the atherogenic one.
The result is a particle that is simultaneously pro-atherogenic, pro-thrombotic, and proinflammatory. It is also particularly strongly associated with aortic valve stenosis — a mechanistically distinct pathway from coronary artery disease that few patients know elevated Lp(a) drives. If I see a patient with elevated Lp(a) and any murmur or valve symptoms, that gets immediate attention.
Can Lp(a) Be Lowered?
The current treatment picture for Lp(a) is nuanced and evolving rapidly:
- Statins do not lower Lp(a) and may slightly raise it — an important point when deciding how aggressively to pursue statin dose escalation in a high-Lp(a) patient who appears otherwise controlled on LDL-C.
- Niacin can reduce Lp(a) by 20–30% but carries an unfavorable side effect profile and has not demonstrated cardiovascular outcome benefit in major clinical trials despite Lp(a) lowering.
- PCSK9 inhibitors (evolocumab, alirocumab) can reduce Lp(a) by 20–30% alongside their primary LDL-C-lowering effect. For high-Lp(a) patients who also have elevated LDL or who are statin-intolerant, PCSK9 inhibitors offer the most practical available Lp(a)-lowering option today.
- RNA-targeting drugs — pelacarsen (an antisense oligonucleotide) and olpasiran (a small interfering RNA) — are currently in late-stage Phase III clinical trials and have demonstrated 80–90% reductions in Lp(a) levels. These are transformative reductions. Cardiovascular outcome data from these trials is expected within the next few years. If those data show that lowering Lp(a) by 80–90% reduces cardiovascular events — which the mechanistic and epidemiological evidence strongly suggests it should — the management of elevated Lp(a) will change completely.
Current clinical approach for high Lp(a): manage all other risk factors as aggressively as possible, consider PCSK9 inhibitor candidacy if LDL-C or overall risk warrants it, eliminate every other controllable cardiovascular risk factor, and watch closely for emerging therapy data. Testing today also positions you to be an early candidate for these drugs when they reach approval.
"When ApoB outperforms LDL-C as a cardiovascular risk marker in 9 of 9 studies, and a landmark 2026 guideline formally endorses ApoB testing — the argument for continuing to use LDL-C alone becomes increasingly difficult to defend."
The Gap in Standard Care
Standard lipid panels measure four things: total cholesterol, LDL-C (often calculated via the Friedewald equation, not directly measured), HDL-C, and triglycerides. That is it. They do not measure ApoB, Lp(a), LDL particle number, or any other direct index of atherogenic particle burden.
The result is a cardiovascular risk assessment that systematically misses:
- Every patient with discordant LDL-C and ApoB — the small, dense LDL pattern driven by insulin resistance and elevated triglycerides, which is increasingly prevalent in the American population
- Every patient with elevated Lp(a) whose LDL-C appears well-controlled — a population that receives false reassurance about their risk
- The residual cardiovascular risk that persists in patients on maximally tolerated statin therapy, a phenomenon that guideline bodies have been grappling with for years and that ApoB measurement directly addresses
The standard lipid panel was designed in an era when LDL-C was the best available measure. Better tools now exist. The argument for not using them is largely inertia and test-ordering convention — not evidence.
How We Use Advanced Lipid Testing at Pravida Health
Advanced lipid panels — including ApoB and Lp(a) alongside the standard lipid panel — are included in comprehensive labwork across all Pravida Health membership tiers. These are not add-ons we order when something looks off. They are standard baseline assessments for every patient, because the information they provide is simply too important to omit.
We don't treat these values in isolation. ApoB is interpreted alongside LDL-C, non-HDL-C, fasting insulin, inflammatory markers (hsCRP, LP-PLA2), and the full metabolic biomarker picture. When ApoB and LDL-C are discordant — when ApoB is elevated despite normal LDL-C — that discordance tells a story. In the majority of cases, it is explained by insulin resistance, elevated triglycerides, and a small-dense LDL particle pattern. Treating the underlying metabolic dysfunction — improving insulin sensitivity, reducing triglycerides through dietary carbohydrate modification and, where indicated, pharmacological support — often corrects the ApoB more effectively than simply escalating statin dosing, and it addresses the actual driving pathology rather than a downstream marker.
Lp(a) is a permanent fixture of every patient's record. It doesn't require repeat testing since it is genetically fixed and will not change meaningfully over time. But knowing it once allows us to calibrate the appropriate aggressiveness of all other cardiovascular risk factor management. A patient with Lp(a) of 40 mg/dL gets a different conversation than one with Lp(a) of 180 mg/dL — even if their LDL-C and blood pressure are identical.
We also incorporate coronary artery calcium (CAC) scoring for patients at intermediate cardiovascular risk. The 2026 ACC/AHA guideline recommends CAC scoring for men over 40 and women over 45 with borderline or intermediate risk — and when combined with ApoB and Lp(a) results, CAC gives us a complete three-dimensional picture of what is actually happening in the coronary vasculature, not just what the lipid panel predicts might be happening.
What You Can Do Today
- Ask for ApoB at your next lipid panel. It costs approximately $15–25 as a standalone add-on and is covered by most insurance for patients with metabolic risk factors. Direct-access laboratories allow self-ordering without a physician referral if your doctor is not yet routinely ordering it.
- Ask for Lp(a) — at least once in your life. The 2026 ACC/AHA guideline now recommends universal testing. This is especially important if you have a family history of heart disease at younger ages, a personal or family history of familial hypercholesterolemia, or any unexplained cardiovascular events despite apparently well-controlled traditional risk factors.
- If your ApoB is elevated, investigate insulin resistance. Elevated ApoB with normal or near-normal LDL-C is frequently associated with small, dense LDL driven by insulin resistance and elevated triglycerides. Add fasting insulin and HOMA-IR to your next lipid panel. The driving pathology is metabolic, and that is where the intervention needs to focus.
- Understand your LDL-C in context. If LDL-C and ApoB agree — both normal or both elevated — LDL-C is a reasonable guide to treatment. If they disagree, ApoB is the more accurate risk indicator. The direction of that disagreement matters clinically and should drive your treatment conversation.
- If Lp(a) is high, manage everything else aggressively. A high Lp(a) is not a reason to panic. It is a reason to have zero remaining controllable cardiovascular risk factors. Optimal LDL-C and ApoB. Optimal blood pressure. No smoking. Metabolic health in order. And watch closely for the RNA-targeting therapy data that may offer a specific solution within the next few years.
Frequently Asked Questions
What is ApoB testing and why is it better than LDL cholesterol?
ApoB directly measures the number of atherogenic lipoprotein particles in your blood — one ApoB molecule per particle. LDL cholesterol measures the cholesterol content of LDL particles, which varies based on particle size. When the two are discordant, ApoB is a more accurate predictor of cardiovascular risk. A 2025 systematic review of 15 studies covering 593,354 participants found ApoB outperformed LDL-C in every discordance analysis.
What is a normal Lp(a) level?
Lp(a) below 30 mg/dL (75 nmol/L) is generally considered low risk. The 2026 ACC/AHA guideline defines 125 nmol/L (approximately 50 mg/dL) as a risk-enhancing threshold, associated with approximately 1.4-fold increased long-term cardiovascular risk. Above 250 nmol/L carries at least twofold increased risk.
Can statins lower Lp(a)?
No — statins do not meaningfully reduce Lp(a) and may slightly increase it. PCSK9 inhibitors can reduce Lp(a) by 20–30%. RNA-targeting drugs in late-stage trials (pelacarsen, olpasiran) reduce Lp(a) by 80–90% and may transform management of this risk factor in the next 2–4 years.
Should everyone get an ApoB test?
The 2026 ACC/AHA guidelines recommend ApoB for patients who have reached LDL-C goals but have residual cardiovascular risk factors — metabolic syndrome, type 2 diabetes, high triglycerides, or known cardiovascular disease. ApoB is particularly valuable in anyone whose LDL-C may underestimate true particle burden.
What is the difference between Lp(a) and LDL cholesterol?
LDL particles carry cholesterol from the liver to tissues and are the primary atherogenic lipoprotein in most adults — modifiable by diet, exercise, and statins. Lp(a) is a distinct lipoprotein with an additional protein (apolipoprotein(a)) that makes it both atherogenic and thrombogenic. Unlike LDL, Lp(a) is approximately 80–90% genetically determined and does not respond meaningfully to lifestyle modification.
Get the Complete Cardiovascular Picture
Your standard lipid panel may be missing your most important cardiovascular risk data. At Pravida Health, advanced lipid testing — including ApoB and Lp(a) — is standard in every comprehensive metabolic assessment.
Schedule a Consultation View Memberships